The fate of pollen in two morphologically contrasting buzz-pollinated Solanum flowers
DOI:
https://doi.org/10.26786/1920-7603(2025)836Keywords:
Buzz pollination, floral morphology, heteranthery, pollen carryover, pollen transfer and deposition, pollen fatesAbstract
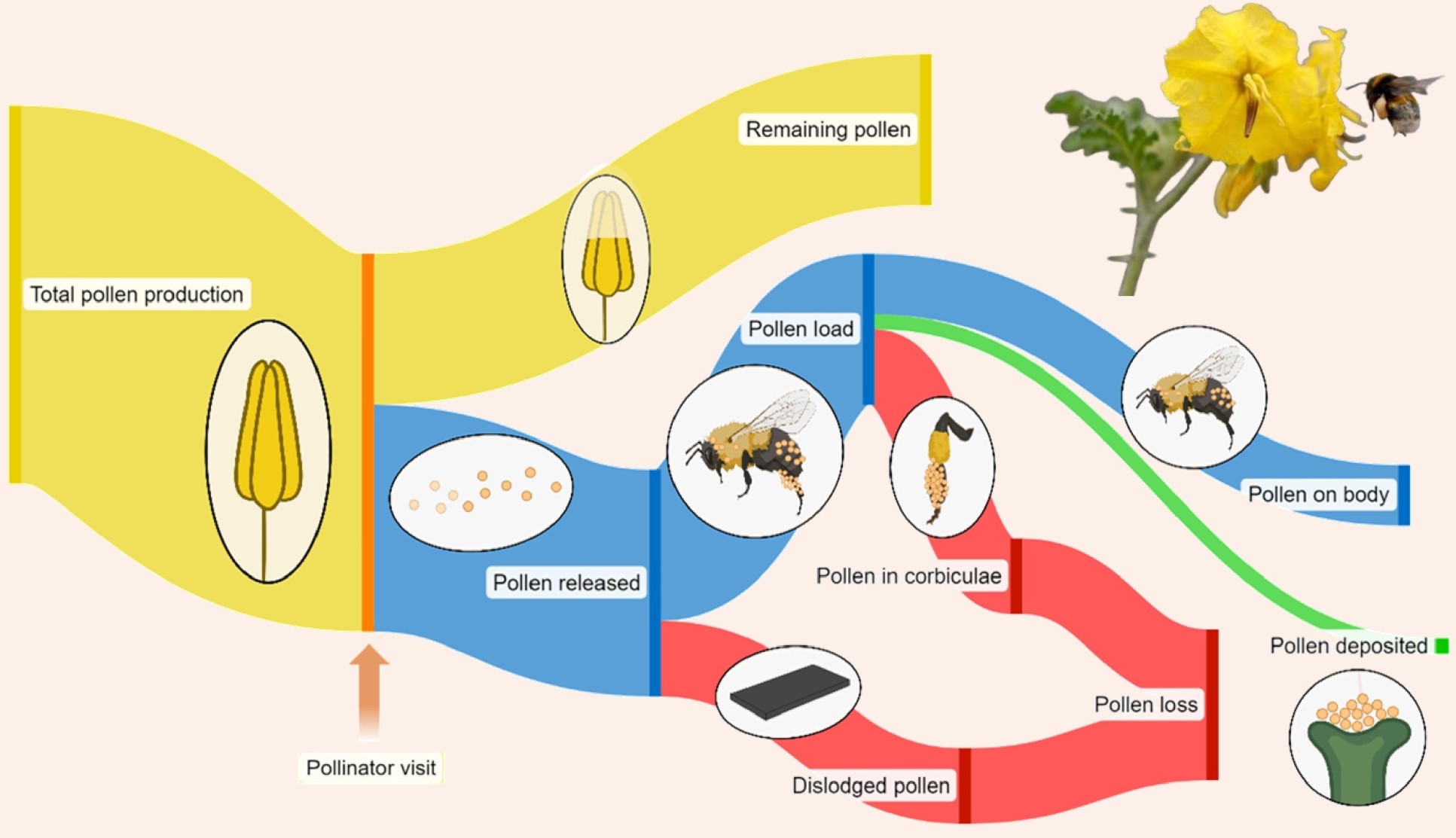
Pollen transfer efficiency (PTE) and pollen deposition patterns on a pollinator’s body significantly influence plant reproductive success. However, studies on pollen fates (i.e., the destination of pollen grains after anther dehiscence) in animal-pollinated species offering pollen as the sole reward are limited. This study investigated pollen fates in two nectarless, buzz-pollinated Solanum species with contrasting floral morphology. Experimental trials were conducted involving one pollen donor and four recipient flowers of Solanum rostratum Dunal or S. dulcamara L., using captive Bombus terrestris L. as pollinators. The number of pollen grains remaining in the anthers, deposited on stigmas, placed on the pollinator, or falling to the ground was quantified. Both species produced a relatively high number of pollen grains as expected for buzz-pollinated plants. Pollen deposition curves followed exponential decay patterns, with a higher number of pollen grains deposited for S. dulcamara, and a similar rate of decline for both species. PTE was similar between species (0.86% vs. 1.00%, for S. rostratum and S. dulcamara, respectively) but these values could be 25% higher if we were to measure pollen deposition in 20 recipients rather than four as in the present study. Although both species had similar PTE values, their pollen fates differed: in S. rostratum, pollen was mainly lost to the ground, whereas in S. dulcamara, it was primarily removed by bee grooming, likely due to differences in their floral architectures. These findings suggest that species with different flower morphology could exhibit different pollen fates without impacting pollen transfer to conspecific stigmas.
References
Bowers KA (1975) The pollination ecology of Solanum rostratum (Solanaceae). American Journal of Botany 62:633–638. DOI: https://doi.org/10.1002/j.1537-2197.1975.tb14094.x
Brooks ME, Kristensen K, van Benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Mächler M, Bolker BM (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. The R Journal 9:378-400. DOI: https://doi.org/10.32614/RJ-2017-066
Buchmann SL (1983) Buzz pollination in angiosperms. In: Jones CE, Little RJ (eds) Handbook of Experimental Pollination Biology. Scientific and Academic Editions, New York, pp 73–113.
Burnham KP, Anderson DR (2002) Model Selection and Multimodel Inference. A Practical Information-Theoretic Approach. SpringerVerlag, New York.
Castellanos MC, Wilson P, Thomson JD (2003) Pollen transfer by hummingbirds and bumblebees, and the divergence of pollination modes in Penstemon. Evolution 57:2742–2752. DOI: https://doi.org/10.1111/j.0014-3820.2003.tb01516.x
Christopher DA, Mitchell RJ, Karron JD (2020) Pollination intensity and paternity in flowering plants. Annals of Botany 125:1–9. DOI: https://doi.org/10.1093/aob/mcz159
Cruden RW (2000) Pollen grains: why so many? In: Dafni A, Hesse M, Pacini E (eds) Pollen and Pollination. Springer, Berlin. DOI: https://doi.org/10.1007/978-3-7091-6306-1_8
De Luca P, Vallejo-Marín M (2013) What’s the ‘buzz’ about? The ecology and evolutionary significance of buzz-pollination. Current Opinion in Plant Biology 16:429–435. DOI: https://doi.org/10.1016/j.pbi.2013.05.002
Dellinger AS, Paun O, Baar J, Temsch EM, Fernández‐Fernández D, Schönenberger J (2022) Population structure in Neotropical plants: integrating pollination biology, topography and climatic niches. Molecular Ecology 31:2264–2280. DOI: https://doi.org/10.1111/mec.16403
Fox J, Weisberg S (2019) car: companion to applied regression. [online] URL: https://CRAN.R-project.org/package=car
Free JB (1970) The flower constancy of bumblebees. Journal of Animal Ecology 39:395–402. DOI: https://doi.org/10.2307/2978
Gamba D, Muchhala N (2020) Global patterns of population genetic differentiation in seed plants. Molecular Ecology 29:3413–3428. DOI: https://doi.org/10.1111/mec.15575
Gamba D, Muchhala N (2023) Pollinator type strongly impacts gene flow within and among plant populations for six Neotropical species. Ecology 104:3845. DOI: https://doi.org/10.1002/ecy.3845
Glover BJ, Bunnewell S, Martin C (2004) Convergent evolution within the genus Solanum: The specialised anther cone develops through alternative pathways. Gene 331:1–7. DOI: https://doi.org/10.1016/j.gene.2004.01.027
Harder LD (1990) Behavioural responses by bumble bees to variation in pollen availability. Oecologia 85:41–47. DOI: https://doi.org/10.1007/BF00317341
Harder LD, Barclay RMR (1994) The functional significance of poricidal anthers and buzz pollination: controlled pollen removal from Dodecatheon. Functional Ecology 8:509–517. DOI: https://doi.org/10.2307/2390076
Harder LD, Thomson JD (1989) Evolutionary options for maximizing pollen dispersal of animal-pollinated plants. The American Naturalist 133:323–344. DOI: https://doi.org/10.1086/284922
Harder LD, Wilson WG (1994) Floral evolution and male reproductive success: Optimal dispensing schedules for pollen dispersal by animal-pollinated plants. Evolutionary Ecology 8:542–559. DOI: https://doi.org/10.1007/BF01238257
Hartig F (2024) DHARMa: residual diagnostics for hierarchical (multi-Level / mixed) regression models. R package version 0.4.7. [online] URL: https://CRAN.R-project.org/package=DHARMa
Hilgenhof R, Gagnon E, Knapp S (2023) Morphological trait evolution in Solanum (Solanaceae): Evolutionary lability of key taxonomic characters. Taxon 72:811–847. DOI: https://doi.org/10.1002/tax.12990
Holmquist KG, Mitchell RJ, Karron JD (2012) Influence of pollinator grooming on pollen‐mediated gene dispersal in Mimulus ringens (Phrymaceae). Plant Species Biology 27:77–85. DOI: https://doi.org/10.1111/j.1442-1984.2011.00329.x
Jesson LK, Barrett SCH (2002) Solving the puzzle of mirror-image flowers. Nature 417:707–707. DOI: https://doi.org/10.1038/417707a
Johnson SD, Harder LD (2023) The economy of pollen dispersal in flowering plants. Proceedings of the Royal Society B: Biological Sciences 290:20231148. DOI: https://doi.org/10.1098/rspb.2023.1148
Johnson SD, Neal PR, Harder, LD (2005). Pollen fates and the limits on male reproductive success in an orchid population. Biological Journal of the Linnean Society, 86(2), 175-190. DOI: https://doi.org/10.1111/j.1095-8312.2005.00541.x
Kearns CA, Inouye DW (1993) Techniques for Pollination Biologists. University press of Colorado, Colorado.
Kemp JE, Vallejo-Marín M (2021) Pollen dispensing schedules in buzz-pollinated plants: experimental comparison of species with contrasting floral morphologies. American Journal of Botany 108:993–1005. DOI: https://doi.org/10.1002/ajb2.1680
Knapp S (2013) A revision of the Dulcamaroid Clade of Solanum L. (Solanaceae). PhytoKeys 22:1–428. DOI: https://doi.org/10.3897/phytokeys.22.4041
Larson BM, Barrett SC (1999) The ecology of pollen limitation in buzz‐pollinated Rhexia virginica (Melastomataceae). Journal of Ecology 87:371–381. DOI: https://doi.org/10.1046/j.1365-2745.1999.00362.x
Lenth RV (2016) Least-squares means: the r package lsmeans. Journal of Statistical Software 69:1–33. DOI: https://doi.org/10.18637/jss.v069.i01
Lertzman KP (1981) Pollen transfer: processes and consequences. University of British Columbia, Vancouver. [online] URL: https://open.library.ubc.ca/collections/ubctheses/831/items/1.0095274
Luo Z, Zhang D, Renner SS (2008) Why two kinds of stamens in buzz-pollinated flowers? Experimental support for Darwin’s division-of-labour hypothesis. Functional Ecology 22:794–800. DOI: https://doi.org/10.1111/j.1365-2435.2008.01444.x
Minnaar C, Anderson B, Jager ML, Karron JD (2019) Plant–pollinator interactions along the pathway to paternity. Annals of Botany 123:225–245. DOI: https://doi.org/10.1093/aob/mcy167
Mitchell RJ, Wilson WG, Holmquist KG, Karron JD (2013) Influence of pollen transport dynamics on sire profiles and multiple paternity in flowering plants. PLoS One 8(10), e76312. DOI: https://doi.org/10.1371/journal.pone.0076312
Moquet L, Bruyère L, Pirard B, Jacquemart AL (2017) Nectar foragers contribute to the pollination of buzz-pollinated plant species. American Journal of Botany 104:1451–1463. DOI: https://doi.org/10.3732/ajb.1700090
Mora-Carrera E, Castañeda-Zárate M, Fornoni J, Boege K, Domínguez CA (2019) On the adaptive value of monomorphic versus dimorphic enantiostyly in Solanum rostratum. Annals of Botany 123:205–212. DOI: https://doi.org/10.1093/aob/mcy162
Morris WF, Price MV, Waser NM, Thomson JD, Thomson B, Stratton DA (1994) Systematic increase in pollen carryover and its consequences for geitonogamy in plant populations. Oikos 71:431–440. DOI: https://doi.org/10.2307/3545831
Nevard L, Vallejo-Marín M (2022) Floral orientation affects outcross-pollen deposition in buzz-pollinated flowers with bilateral symmetry. American Journal of Botany 109:1568–1578. DOI: https://doi.org/10.1002/ajb2.16078
Posit team (2025). RStudio: Integrated Development Environment for R. Posit Software, PBC, Boston, MA. [online] URL: http://www.posit.co/
Price MV, Waser NM (1982) Experimental studies of pollen carryover: hummingbirds and Ipomopsis aggregata. Oecologia 54:353–358. DOI: https://doi.org/10.1007/BF00380004
R Core Team (2024) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. [online] URL: https://www.R-project.org/
Rademaker MCJ, De Jong TJ, & Klinkhamer PGL (1997). Pollen dynamics of bumble-bee visitation on Echium vulgare. Functional Ecology, 11(5), 554-563. DOI: https://doi.org/10.1046/j.1365-2435.1997.00124.x
Richards SA, Williams NM, Harder LD (2009) Variation in pollination: causes and consequences for plant reproduction. The American Naturalist 174:382–398. DOI: https://doi.org/10.1086/603626
Robertson AW (1992) The relationship between floral display size, pollen carryover and geitonogamy in Myosotis colensoi (Kirk) Macbride (Boraginaceae). Biological Journal of the Linnean Society 46:333–349. DOI: https://doi.org/10.1111/j.1095-8312.1992.tb00868.x
Russell AL, Buchmann SL, Papaj DR (2017) How a generalist bee achieves high efficiency of pollen collection on diverse floral resources. Behavioral Ecology 28:991–1003. DOI: https://doi.org/10.1093/beheco/arx058
Santa-Martinez E, Cardoso Castro C, Flick A (2021) Bee species visiting Medicago sativa differ in pollen deposition curves with consequences for gene flow. American Journal of Botany 108:1016–1028. DOI: https://doi.org/10.1002/ajb2.1683
Snow AA, Roubik DW (1987) Pollen deposition and removal by bees visiting two tree species in Panama. Biotropica 19:57–63. DOI: https://doi.org/10.2307/2388460
Solís-Montero L, Vallejo-Marín M (2017) Does the morphological fit between flowers and pollinators affect pollen deposition? An experimental test in a buzz-pollinated species with anther dimorphism. Ecology and Evolution 7:2706–2715. DOI: https://doi.org/10.1002/ece3.2897
Solís-Montero L, Vergara CH, Vallejo-Marín M (2015) High incidence of pollen theft in natural populations of a buzz-pollinated plant. Arthropod-Plant Interactions 9:599–611. DOI: https://doi.org/10.1007/s11829-015-9397-5
Thomson JD (1986) Pollen transport and deposition by bumble bees in Erythronium: influences of floral nectar and bee grooming. Journal of Ecology 74: 329-341. DOI: https://doi.org/10.2307/2260258
Thomson JD (2006) Tactics for male reproductive success in plants: contrasting insights of sex allocation theory and pollen presentation theory. Integrative and Comparative Biology 46:390–397. DOI: https://doi.org/10.1093/icb/icj046
Thomson JD, Goodell K (2001) Pollen removal and deposition by honeybee and bumblebee visitors to apple and almond flowers. Journal of Applied Ecology 38:1032–1044. DOI: https://doi.org/10.1046/j.1365-2664.2001.00657.x
Thomson JD, Plowright RC (1980) Pollen carryover, nectar rewards, and pollinator behaviour with special reference to Diervilla lonicera. Oecologia 46:68–74. DOI: https://doi.org/10.1007/BF00346968
Thomson JD, Wilson P, Valenzuela M, Malzone M. (2000). Pollen presentation and pollination syndromes, with special reference to Penstemon. Plant Species Biology, 15(1), 11-29. DOI: https://doi.org/10.1046/j.1442-1984.2000.00026.x
Thorp RW (2000). The collection of pollen by bees. In: Dafni, A., Hesse, M., Pacini, E. (eds) Pollen and Pollination. Springer, Vienna. DOI: https://doi.org/10.1007/978-3-7091-6306-1_11
Thøstesen AM, Olesen JM (1996) Pollen removal and deposition by specialist and generalist bumblebees in Aconitum septentrionale. Oikos 77:77–84. DOI: https://doi.org/10.2307/3545587
Vallejo-Marín M, Manson JS, Thomson JD, Barrett SCH (2009) Division of labour within flowers: heteranthery, a floral strategy to reconcile contrasting pollen fates. Journal of Evolutionary Biology 22:828–839. DOI: https://doi.org/10.1111/j.1420-9101.2009.01693.x
Vallejo-Marín M, Pereira Nunes CE, Russell AL (2022) Anther cones increase pollen release in buzz-pollinated Solanum flowers. Evolution 76:931–945. DOI: https://doi.org/10.1111/evo.14485
Vallejo-Marín M, Russell AL (2024) Harvesting pollen with vibrations: towards an integrative understanding of the proximate and ultimate reasons for buzz pollination. Annals of Botany 133:379–398. DOI: https://doi.org/10.1093/aob/mcad189
Vallejo-Marín M, Solís-Montero L, Souto Vilaros D, Lee MYQ (2013) Mating system in Mexican populations of the annual herb Solanum rostratum Dunal (Solanaceae). Plant Biology 15:948–954. DOI: https://doi.org/10.1111/j.1438-8677.2012.00715.x
Vallejo-Marín M, Walker C, Friston-Reilly P, Solís-Montero L, Igic B (2014) Recurrent modification of floral morphology in heterantherous Solanum reveals a parallel shift in reproductive strategy. Philosophical Transactions of the Royal Society B: Biological Sciences 369:20130256. DOI: https://doi.org/10.1098/rstb.2013.0256
Zhang L, Yu L, Lou A (2017) No evolutionary change in the mating system of Solanum rostratum (Solanaceae) during its invasion in China. Scientific Reports 7:17698. DOI: https://doi.org/10.1038/s41598-017-17881-2
Zhao J, Solís-Montero L, Lou A, Vallejo-Marín M (2013) Population structure and genetic diversity of native and invasive populations of Solanum rostratum (Solanaceae). PLOS ONE 8:79807. DOI: https://doi.org/10.1371/journal.pone.0079807
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 Christian Ary Vasquez-Castro, Elodie Morel, Bernardo Garcia-Simpson, Mario Vallejo-Marín

This work is licensed under a Creative Commons Attribution 4.0 International License.












