Long-term effects of nitrogen enrichment in pollen chemistry of a plant species from Brazilian savannas, Pavonia rosa campestris
DOI:
https://doi.org/10.26786/1920-7603(2023)756Keywords:
Brazilian savannas, global changes, pollen chemistry, soil eutrophicationAbstract
Human activities have substantially increased soil nutrient availability during the past decades, affecting plant community composition and plants' nutritional content. Several amino acids found in pollen, the main source of larval diet for bees, affect the development, health, and behaviour of this important group of pollinators. A better understanding of the consequences of global changes on pollen amino acid content can help explain and predict future impacts on bee populations and diversity. This is particularly relevant in regions that are highly exposed to fertilizers due to their importance for global food production, such as the Brazilian savannas (Cerrado), where soils are mostly dystrophic.
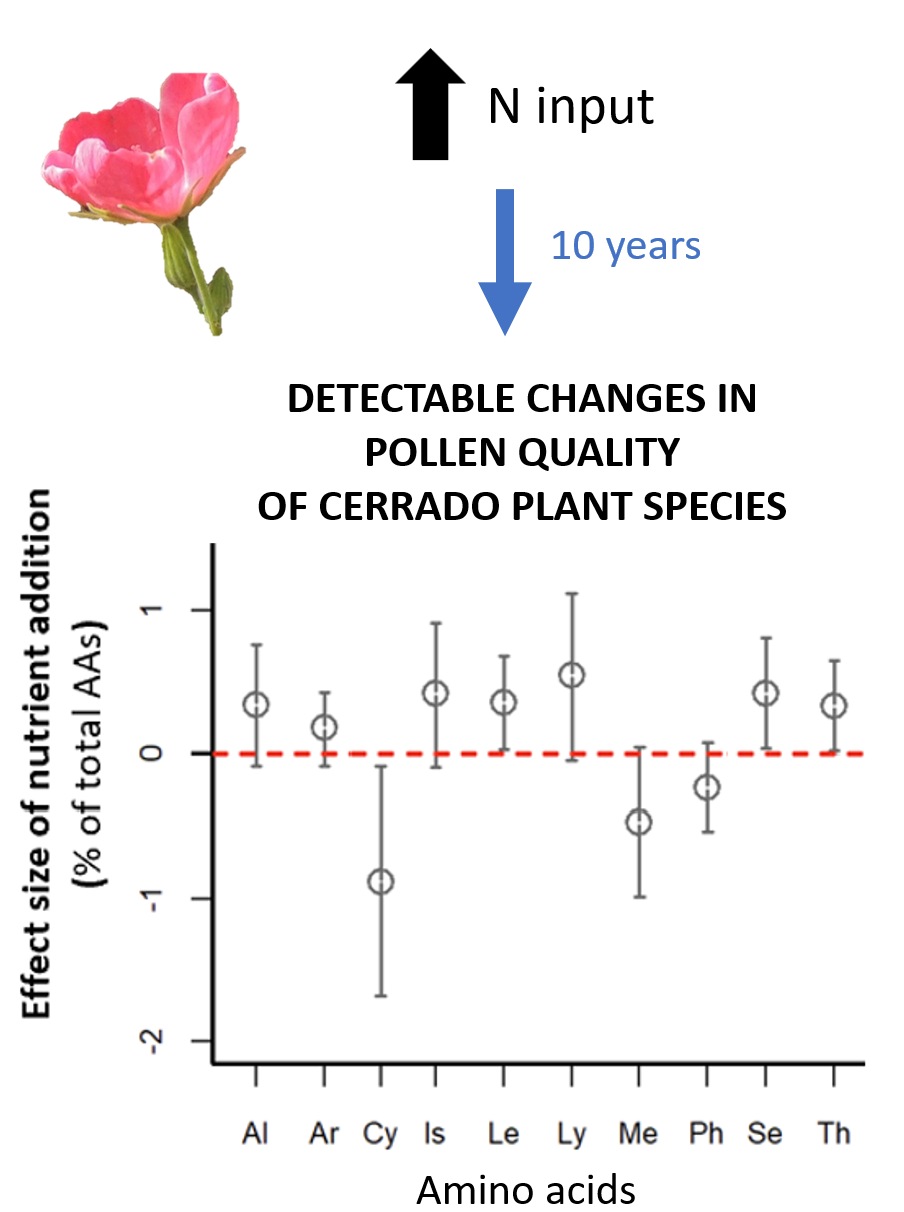
Here, we use a long-term controlled fertilization experiment conducted in Cerrado and demonstrate that even after 10 years since the last fertilization addition, effects were still detectable on leaf and pollen chemical content. More specifically, pollen amino acid content of Pavonia rosa-campestris (Malvaceae), a species known to be important for the diet of several native bee species, changed because of nitrogen (N) addition. Not only did the overall amino acid content increase with N addition, but its profile was also affected, with the proportion of some amino acids increasing (e.g. isoleucine, leucine, serine, threonine), while decreasing for others (e.g. cysteine).
These amino acids can have important effects on larval development and flower visitor behaviour. Further studies evaluating the effects on a diverse set of plant species and the consequent impacts on flower visitation and bee fitness are essential to better understand the full consequences of increased nitrogen availability in nutrient-limited ecosystems such as Cerrado.
References
Adler LS, Irwin RE (2005) Ecological costs and benefits of defenses in nectar. Ecology 86:2968-2978. https://doi.org/10.1890/05-0118 DOI: https://doi.org/10.1890/05-0118
Alm J, Ohnmeiss TE, Lanza J, Vriesenga L (1990) Preference of cabbage white butterflies and honey bees for nectar that contains amino acids. Oecologia 84:53-57. https://doi.org/10.1007/BF00665594 DOI: https://doi.org/10.1007/BF00665594
AOAC - Association of Official Analytical Chemists (1980) Official Methods of Analysis, AOAC, Washington, DC, USA.
Barbosa ERM, van Langevelde F, Tomlinson KW, Carvalheiro LG, Kirkman K, de Bie S, Prins HHT (2014) Tree species from different functional groups respond differently to environmental changes during establishment. Oecologia 174:1345-1357. https://doi.org/10.1007/s00442-013-2853-y DOI: https://doi.org/10.1007/s00442-013-2853-y
Barbosa ERM, Tomlinson KW, Carvalheiro LG, Kirkman K, de Bie S, Prins HHT, van Langevelde F (2014) Short-Term Effect of Nutrient Availability and Rainfall Distribution on Biomass Production and Leaf Nutrient Content of Savanna Tree Species Bond-Lamberty B (ed). PLoS ONE 9:e92619. https://doi.org/10.1371/journal.pone.0092619 DOI: https://doi.org/10.1371/journal.pone.0092619
Bobbink R, Hicks K, Galloway J, Spranger T, Alkemade R, Ashmore M, Bustamante M, Cinderby S, Davidson E, Dentener F, Emmett B, Erisman J-W, Fenn M, Gilliam F, Nordin A, Pardo L, De Vries W (2010) Global assessment of nitrogen deposition effects on terrestrial plant diversity: a synthesis. Ecological Applications 20:30-59. https://doi.org/10.1890/08-1140.1 DOI: https://doi.org/10.1890/08-1140.1
Bonoan RE, Gonzalez J, Starks PT (2020) The perils of forcing a generalist to be a specialist: lack of dietary essential amino acids impacts honey bee pollen foraging and colony growth. Journal of Apicultural Research 59:95-103. https://doi.org/10.1080/00218839.2019.1656702 DOI: https://doi.org/10.1080/00218839.2019.1656702
Bouwman AF, Beusen AHW, Lassaletta L, van Apeldoorn DF, van Grinsven HJM, Zhang J, Ittersum van MK (2017) Lessons from temporal and spatial patterns in global use of N and P fertilizer on cropland. Scientific Reports 7:40366. https://doi.org/10.1038/srep40366 DOI: https://doi.org/10.1038/srep40366
Brodbeck, B SD Amino acid nutrition of herbivorous insects and stress to host plants. In: Insects outbreaks.
Buttrose M, Grant W, Lott J (1977) Reversible curvature of style branches of Hibiscus trionum L., a pollination mechanism. Australian Journal of Botany 25:567. https://doi.org/10.1071/BT9770567 DOI: https://doi.org/10.1071/BT9770567
Carlesso D, Smargiassi S, Pasquini E, Bertelli G, Baracchi D (2021) Nectar non-protein amino acids (NPAAs) do not change nectar palatability but enhance learning and memory in honey bees. Scientific Reports 11:11721. https://doi.org/10.1038/s41598-021-90895-z DOI: https://doi.org/10.1038/s41598-021-90895-z
Ceulemans T, Hulsmans E, Vanden Ende W, Honnay O (2017) Nutrient enrichment is associated with altered nectar and pollen chemical composition in Succisa pratensis Moench and increased larval mortality of its pollinator Bombus terrestris L. Nascimento FS (ed). PLOS ONE 12:e0175160. https://doi.org/10.1371/journal.pone.0175160 DOI: https://doi.org/10.1371/journal.pone.0175160
Chang CL (2004) Effect of Amino Acids on Larvae and Adults of Ceratitis capitata (Diptera: Tephritidae). Annals of the Entomological Society of America 97:529-535. https://doi.org/10.1603/0013-8746(2004)097[0529:EOAAOL]2.0.CO;2 DOI: https://doi.org/10.1603/0013-8746(2004)097[0529:EOAAOL]2.0.CO;2
Cook SM, Awmack CS, Murray DA, Williams IH (2003) Are honey bees' foraging preferences affected by pollen amino acid composition? Ecological Entomology 28: 622-627. https://doi.org/10.1046/j.1365-2311.2003.00548.x DOI: https://doi.org/10.1046/j.1365-2311.2003.00548.x
Cook D, Manson JS, Gardner DR, Welch KD, Irwin RE (2013) Norditerpene alkaloid concentrations in tissues and floral rewards of larkspurs and impacts on pollinators. Biochemical Systematics and Ecology 48:123-131. https://doi.org/10.1016/j.bse.2012.11.015 DOI: https://doi.org/10.1016/j.bse.2012.11.015
David TI, Storkey J, Stevens CJ (2019) Understanding how changing soil nitrogen affects plant-pollinator interactions. Arthropod-Plant Interactions 13:671-684. https://doi.org/10.1007/s11829-019-09714-y DOI: https://doi.org/10.1007/s11829-019-09714-y
de Mello TRB, Munhoz CBR, Bustamante MMC (2014) Effects of nutrient additions on the diversity of the herbaceous-subshrub layer of a Brazilian savanna (Cerrado). In: Sutton MA, Mason KE, Sheppard LJ, Sverdrup H, Haeuber R, Hicks WK (eds) Nitrogen deposition, critical loads and biodiversity. Springer, Dordrecht, pp 147-154. https://doi.org/10.1007/978-94-007-7939-6_16 DOI: https://doi.org/10.1007/978-94-007-7939-6_16
Domingos-Melo A, Bezerra SM da S, Nadia T de L, Machado IC (2020) The dark side of the rain: self-pollination setbacks due to water exposure in Pavonia varians Moric (Malvaceae), a species with rain-dependent flowering. Acta Botanica Brasilica 34:437-441. https://doi.org/10.1590/0102-33062020abb0023 DOI: https://doi.org/10.1590/0102-33062020abb0023
Falkowski PG, Fenchel T, Delong EF (2008) The microbial engines that drive Earth's biogeochemical cycles. Science 320:1034-1039. https://doi.org/10.1126/science.1153213 DOI: https://doi.org/10.1126/science.1153213
FAO/IIASA/ISRIC/ISS-CAS/JRC, 2012. Harmonized World Soil Database (version 1.2). FAO, Rome, Italy and IIASA, Laxenburg, Austria
Finkelstein CJ, CaraDonna PJ, Gruver A, Welti EAR, Kaspari M, Sanders NJ (2022) Sodium-enriched floral nectar increases pollinator visitation rate and diversity. Biology Letters 18:20220016. https://doi.org/10.1098/rsbl.2022.0016 DOI: https://doi.org/10.1098/rsbl.2022.0016
Gardener MC, Gillman MP (2001a) Analyzing Variability in Nectar Amino Acids: Composition Is Less Variable Than Concentration. Journal of Chemical Ecology 27:2545-2558. https://doi.org/10.1023/A:1013687701120 DOI: https://doi.org/10.1023/A:1013687701120
Gardener MC, Gillman MP (2001b) The effects of soil fertilizer on amino acids in the floral nectar of corncockle, Agrostemma githago (Caryophyllaceae). Oikos 92:101-106. https://doi.org/10.1034/j.1600-0706.2001.920112.x DOI: https://doi.org/10.1034/j.1600-0706.2001.920112.x
Gardener MC, Gillman MP (2002) The taste of nectar - a neglected area of pollination ecology. Oikos 98:552-557. https://doi.org/10.1034/j.1600-0706.2002.980322.x DOI: https://doi.org/10.1034/j.1600-0706.2002.980322.x
Gijbels P, Ceulemans T, Van den Ende W, Honnay O (2015) Experimental fertilization increases amino acid content in floral nectar, fruit set and degree of selfing in the orchid Gymnadenia conopsea. Oecologia 179:785-795. https://doi.org/10.1007/s00442-015-3381-8 DOI: https://doi.org/10.1007/s00442-015-3381-8
Gijbels P, Van den Ende W, Honnay O (2014) Landscape scale variation in nectar amino acid and sugar composition in a Lepidoptera pollinated orchid species and its relation with fruit set. Journal of Ecology 102:136-144. https://doi.org/10.1111/1365-2745.12183 DOI: https://doi.org/10.1111/1365-2745.12183
Glavinic U, Stankovic B, Draskovic V, Stevanovic J, Petrovic T, Lakic N, Stanimirovic Z (2017) Dietary amino acid and vitamin complex protects honey bee from immunosuppression caused by Nosema ceranae Jadhao SB (ed). PLOS ONE 12:e0187726. https://doi.org/10.1371/journal.pone.0187726 DOI: https://doi.org/10.1371/journal.pone.0187726
Goodwillie C, Weber JJ (2018) The best of both worlds? A review of delayed selfing in flowering plants. American Journal of Botany 105:641-655. https://doi.org/10.1002/ajb2.1045 DOI: https://doi.org/10.1002/ajb2.1045
Gosselin M, Michez D, Vanderplanck M, Roelants D, Glauser G, Rasmont P (2013) Does Aconitum septentrionale chemically protect floral rewards to the advantage of specialist bumblebees?: Impact of toxic rewards on visitors. Ecological Entomology 38:400-407. https://doi.org/10.1111/een.12032 DOI: https://doi.org/10.1111/een.12032
de Groot AP (1952) Amino acid requirements for growth of the honeybee (Apis mellifica L.). Experientia 8:192-194. https://doi.org/10.1007/BF02173740 DOI: https://doi.org/10.1007/BF02173740
Haddad NM, Haarstad J, Tilman D (2000) The effects of long-term nitrogen loading on grassland insect communities. Oecologia 124:73-84. https://doi.org/10.1007/s004420050026 DOI: https://doi.org/10.1007/s004420050026
Hanley ME, Franco M, Pichon S, Darvill B, Goulson D (2008) Breeding system, pollinator choice and variation in pollen quality in British herbaceous plants. Functional Ecology 22:592-598. https://doi.org/10.1111/j.1365-2435.2008.01415.x DOI: https://doi.org/10.1111/j.1365-2435.2008.01415.x
Haridasan, M (2008) Nutritional adaptations of native plants of the cerrado biome in acid soils. Brazilian Journal of Plant Physiology 20:183-195. https://doi.org/10.1590/S1677-04202008000300003 DOI: https://doi.org/10.1590/S1677-04202008000300003
Heil M (2011) Nectar: generation, regulation and ecological functions. Trends in Plant Science 16:191-200. https://doi.org/10.1016/j.tplants.2011.01.003 DOI: https://doi.org/10.1016/j.tplants.2011.01.003
Hoover SER, Ladley JJ, Shchepetkina AA, Tisch M, Gieseg SP, Tylianakis JM (2012) Warming, CO2, and nitrogen deposition interactively affect a plant-pollinator mutualism: Disruption of plant-pollinator mutualism. Ecology Letters 15:227-234. https://doi.org/10.1111/j.1461-0248.2011.01729.x DOI: https://doi.org/10.1111/j.1461-0248.2011.01729.x
Hrassnigg N, Leonhard B, Crailsheim K (2003) Free amino acids in the haemolymph of honey bee queens (Apis mellifera L.). Amino Acids 24:205-212. https://doi.org/10.1007/s00726-002-0311-y DOI: https://doi.org/10.1007/s00726-002-0311-y
Huang Z (2012) Pollen nutrition affects honey bee stress resistance. Terrestrial Arthropod Reviews 5:175-189. https://doi.org/10.1163/187498312X639568 DOI: https://doi.org/10.1163/187498312X639568
Human H, Nicolson SW, Strauss K, Pirk CWW, Dietemann V (2007) Influence of pollen quality on ovarian development in honeybee workers (Apis mellifera scutellata). Journal of Insect Physiology 53: 649-655. https://doi.org/10.1016/j.jinsphys.2007.04.002 DOI: https://doi.org/10.1016/j.jinsphys.2007.04.002
Inouye DW, Waller GD (1984) Responses of Honey Bees (Apis Mellifera) to Amino Acid Solutions Mimicking Floral Nectars. Ecology 65:618-625. https://doi.org/10.2307/1941424 DOI: https://doi.org/10.2307/1941424
Isbell F, Reich PB, Tilman D, Hobbie SE, Polasky S, Binder S (2013) Nutrient enrichment, biodiversity loss, and consequent declines in ecosystem productivity. Proceedings of the National Academy of Sciences 110:11911-11916. https://doi.org/10.1073/pnas.1310880110 DOI: https://doi.org/10.1073/pnas.1310880110
Jeannerod L, Carlier A, Schatz B, Daise C, Richel A, Agnan Y, Baude M, Jacquemart A-L (2022) Some bee-pollinated plants provide nutritionally incomplete pollen amino acid resources to their pollinators. PLOS ONE 17:e0269992. https://doi.org/10.1371/journal.pone.0269992 DOI: https://doi.org/10.1371/journal.pone.0269992
Kaiser K, Wemheuer B, Korolkow V, Wemheuer F, Nacke H, Schöning I, Schrumpf M, Daniel R (2016) Driving forces of soil bacterial community structure, diversity, and function in temperate grasslands and forests. Scientific Reports 6:33696. https://doi.org/10.1038/srep33696 DOI: https://doi.org/10.1038/srep33696
Linander N, Hempel de Ibarra N, Laska M (2012) Olfactory detectability of L-amino acids in the european honeybee (Apis mellifera), Chemical Senses 37: 631-638. https://doi.org/10.1093/chemse/bjs044 DOI: https://doi.org/10.1093/chemse/bjs044
Lim S, Jung J, Yunusbaev U, Ilyasov R, Kwon HW (2019) Characterization and its implication of a novel taste receptor detecting nutrients in the honey bee, Apis mellifera. Scientific Reports 9:11620. https://doi.org/10.1038/s41598-019-46738-z DOI: https://doi.org/10.1038/s41598-019-46738-z
Lohaus G, Schwerdtfeger M (2014) Comparison of Sugars, Iridoid Glycosides and Amino Acids in Nectar and Phloem Sap of Maurandya barclayana, Lophospermum erubescens, and Brassica napus Kusano M (ed). PLoS ONE 9:e87689. https://doi.org/10.1371/journal.pone.0087689 DOI: https://doi.org/10.1371/journal.pone.0087689
Lošák T, Hlusek J, Martinec J, Vollmann J, Peterka J, Filipcik R, Varga L, Ducsay L, Martensson A (2011) Effect of combined nitrogen and sulphur fertilization on yield and qualitative parameters of Camelina sativa [L.] Crtz. (false flax). Acta Agriculturae Scandinavica, Section B - Soil & Plant Science 61:313-321. https://doi.org/10.1080/09064710.2010.490234 DOI: https://doi.org/10.1080/09064710.2010.490234
Majetic CJ, Fetters AM, Beck OM, Stachnik EF, Beam KM (2017) Petunia floral trait plasticity in response to soil nitrogen content and subsequent impacts on insect visitation. Flora 232:183-193. https://doi.org/10.1016/j.flora.2016.08.002 DOI: https://doi.org/10.1016/j.flora.2016.08.002
Manson JS, Rasmann S, Halitschke R, Thomson JD, Agrawal AA (2012) Cardenolides in nectar may be more than a consequence of allocation to other plant parts: a phylogenetic study of Asclepias Johnson M (ed). Functional Ecology 26:1100-1110. https://doi.org/10.1111/j.1365-2435.2012.02039.x DOI: https://doi.org/10.1111/j.1365-2435.2012.02039.x
Marini L, Tamburini G, Petrucco-Toffolo E, Lindström SAM, Zanetti F, Mosca G, Bommarco R (2015) Crop management modifies the benefits of insect pollination in oilseed rape. Agriculture, Ecosystems & Environment 207:61-66. https://doi.org/10.1016/j.agee.2015.03.027 DOI: https://doi.org/10.1016/j.agee.2015.03.027
McDade LA, Davidar P (1984) Determinants of fruit and seed set in Pavonia dasypetala (Malvaceae). Oecologia 64:61-67. https://doi.org/10.1007/BF00377544 DOI: https://doi.org/10.1007/BF00377544
Midolo G, Alkemade R, Schipper AM, Benítez‐López A, Perring MP, De Vries W (2019) Impacts of nitrogen addition on plant species richness and abundance: A global meta‐analysis Xu X (ed). Global Ecology and Biogeography 28:398-413. https://doi.org/10.1111/geb.12856 DOI: https://doi.org/10.1111/geb.12856
Nepi M (2014) Beyond nectar sweetness: the hidden ecological role of non-protein amino acids in nectar. Journal of Ecology 102:108-115. https://doi.org/10.1111/1365-2745.12170 DOI: https://doi.org/10.1111/1365-2745.12170
Nicholls E, de Ibarra NH (2017). Assessment of pollen rewards by foraging bees. Functional Ecology 31:76-87. https://doi.org/10.1111/1365-2435.12778 DOI: https://doi.org/10.1111/1365-2435.12778
Oksanen J, Simpson G, Blanchet F, Kindt R, Legendre P, Minchin P, O'Hara R, Solymos P, Stevens M, Szoecs E, Wagner H, Barbour M, Bedward M, Bolker B, Borcard D, Carvalho G, Chirico M, De Caceres M, Durand S, Evangelista H, FitzJohn R, Friendly M, Furneaux B, Hannigan G, Hill M, Lahti L, McGlinn D, Ouellette M, Ribeiro Cunha E, Smith T, Stier A, Ter Braak C, Weedon J (2022). vegan: Community Ecology Package_. R package version 2.6-2, https://CRAN.R-project.org/package=vegan.
Ollerton J, Winfree R, Tarrant S (2011) How many flowering plants are pollinated by animals? Oikos 120:321-326. https://doi.org/10.1111/j.1600-0706.2010.18644.x DOI: https://doi.org/10.1111/j.1600-0706.2010.18644.x
Paoli PP, Wakeling LA, Wright GA, Ford D (2014) The dietary proportion of essential amino acids and Sir2 influence lifespan in the honeybee. AGE 36:9649. https://doi.org/10.1007/s11357-014-9649-9 DOI: https://doi.org/10.1007/s11357-014-9649-9
Petanidou T, Van Laere A, N. Ellis W, Smets E (2006) What shapes amino acid and sugar composition in Mediterranean floral nectars? Oikos 115:155-169. https://doi.org/10.1111/j.2006.0030-1299.14487.x DOI: https://doi.org/10.1111/j.2006.0030-1299.14487.x
Pompermaier VT, Campani AR, Dourado E, Coletta LD, Bustamante MM da C, Nardoto GB (2022) Soil mesofauna drives litter decomposition under combined nitrogen and phosphorus additions in a Brazilian woodland savanna. Austral Ecology 47:26-34. https://doi.org/10.1111/aec.13082 DOI: https://doi.org/10.1111/aec.13082
Pöyry J, Carvalheiro LG, Heikkinen RK, Kühn I, Kuussaari M, Schweiger O, Valtonen A, van Bodegom PM, Franzén M (2017) The effects of soil eutrophication propagate to higher trophic levels: Effects of soil eutrophication on herbivores. Global Ecology and Biogeography 26:18-30. https://doi.org/10.1111/geb.12521 DOI: https://doi.org/10.1111/geb.12521
R Core Team (2022). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/.
Rader R, Bartomeus I, Garibaldi LA, Garratt MPD, Howlett BG, Winfree R, Cunningham SA, Mayfield MM, Arthur AD, Andersson GKS, Bommarco R, Brittain C, Carvalheiro LG, Chacoff NP, Entling MH, Foully B, Freitas BM, Gemmill-Herren B, Ghazoul J, Griffin SR, Gross CL, Herbertsson L, Herzog F, Hipólito J, Jaggar S, Jauker F, Klein A-M, Kleijn D, Krishnan S, Lemos CQ, Lindström SAM, Mandelik Y, Monteiro VM, Nelson W, Nilsson L, Pattemore DE, de O. Pereira N, Pisanty G, Potts SG, Reemer M, Rundlöf M, Sheffield CS, Scheper J, Schüepp C, Smith HG, Stanley DA, Stout JC, Szentgyörgyi H, Taki H, Vergara CH, Viana BF, Woyciechowski M (2016) Non-bee insects are important contributors to global crop pollination. Proceedings of the National Academy of Sciences 113:146-151. https://doi.org/10.1073/pnas.1517092112 DOI: https://doi.org/10.1073/pnas.1517092112
Ramos D de L, Bustamante MMC, Silva FD da S e, Carvalheiro LG (2018) Crop fertilization affects pollination service provision - Common bean as a case study Saunders ME (ed). PLOS ONE 13:e0204460. https://doi.org/10.1371/journal.pone.0204460 DOI: https://doi.org/10.1371/journal.pone.0204460
Ribeiro MHM, de Albuquerque PMC, da Luz CFP (2016) Pollen profile of Geopropolis samples collected of "Tiúba" (Melipona (Melikerria) fasciculata Smith 1854) in areas of Cerrado and flooded fields in the state of Maranhão, Brazil. Brazilian Journal of Botany 39:895-912. https://doi.org/10.1007/s40415-016-0280-0 DOI: https://doi.org/10.1007/s40415-016-0280-0
Ruedenauer FA, Leonhardt SD, Lunau K, Spaethe J (2019) Bumblebees are able to perceive amino acids via chemotactile antennal stimulation. Journal of Comparative Physiology A 205:321-331. https://doi.org/10.1007/s00359-019-01321-9 DOI: https://doi.org/10.1007/s00359-019-01321-9
Ruedenauer FA, Spaethe J, Leonhardt SD (2015) How to know which food is good for you: bumblebees use taste to discriminate between different concentrations of food differing in nutrient content. Journal of Experimental Biology 218:2233-2240. https://doi.org/10.1242/jeb.118554 DOI: https://doi.org/10.1242/jeb.118554
Schlindwein C, Martins CF (2000) Competition between the oligolectic bee Ptilothrix plumata (Anthophoridae) and the flower closing beetle Pristimerus calcaratus (Curculionidae) for floral resources of Pavonia cancellata (Malvaceae). Plant Systematics and Evolution 224:183-194. https://doi.org/10.1007/BF00986342 DOI: https://doi.org/10.1007/BF00986342
Schlindwein C, Pick RA, Martins CF (2009) Evaluation of oligolecty in the Brazilian bee Ptilothrix plumata (Hymenoptera, Apidae, Emphorini). Apidologie 40:106-116. https://doi.org/10.1051/apido/2008067 DOI: https://doi.org/10.1051/apido/2008067
Sheng M, Tang J, Yang D, Fisher JB, Wang H, Kattge J (2021) Long-term leaf C:N ratio change under elevated CO2 and nitrogen deposition in China: Evidence from observations and process-based modeling. Science of The Total Environment 800:149591. https://doi.org/10.1016/j.scitotenv.2021.149591 DOI: https://doi.org/10.1016/j.scitotenv.2021.149591
Shiraishi A, Kuwabara M (1970) The Effects of Amino Acids on the Labellar Hair Chemosensory Cells of the Fly. Journal of General Physiology 56:768-782. https://doi.org/10.1085/jgp.56.6.768 DOI: https://doi.org/10.1085/jgp.56.6.768
Silveira R, de Mello T de RB, Silva MRSS, Krüger RH, Bustamante MM da C (2021) Long-term liming promotes drastic changes in the composition of the microbial community in a tropical savanna soil. Biology and Fertility of Soils 57:31-46. https://doi.org/10.1007/s00374-020-01504-6 DOI: https://doi.org/10.1007/s00374-020-01504-6
Simkin SM, Allen EB, Bowman WD, Clark CM, Belnap J, Brooks ML, Cade BS, Collins SL, Geiser LH, Gilliam FS, Jovan SE, Pardo LH, Schulz BK, Stevens CJ, Suding KN, Throop HL, Waller DM (2016) Conditional vulnerability of plant diversity to atmospheric nitrogen deposition across the United States. Proceedings of the National Academy of Sciences 113:4086-4091. https://doi.org/10.1073/pnas.1515241113 DOI: https://doi.org/10.1073/pnas.1515241113
Stabler D, Power EF, Borland AM, Barnes JD, Wright GA (2018) A method for analysing small samples of floral pollen for free and protein‐bound amino acids Carvalheiro L (ed). Methods in Ecology and Evolution 9:430-438. https://doi.org/10.1111/2041-210X.12867 DOI: https://doi.org/10.1111/2041-210X.12867
Steffen W, Richardson K, Rockström J, Cornell SE, Fetzer I, Bennett EM, Biggs R, Carpenter SR, de Vries W, de Wit CA, Folke C, Gerten D, Heinke J, Mace GM, Persson LM, Ramanathan V, Reyers B, Sörlin S (2015) Planetary boundaries: Guiding human development on a changing planet. Science 347:1259855. https://doi.org/10.1126/science.1259855 DOI: https://doi.org/10.1126/science.1259855
Su Y, Ma X, Gong Y, Li K, Han W, Liu X (2021) Responses and drivers of leaf nutrients and resorption to nitrogen enrichment across northern China's grasslands: A meta-analysis. CATENA 199:105110. https://doi.org/10.1016/j.catena.2020.105110 DOI: https://doi.org/10.1016/j.catena.2020.105110
Teulier L, Weber J-M, Crevier J, Darveau C-A (2016) Proline as a fuel for insect flight: enhancing carbohydrate oxidation in hymenopterans. Proceedings of the Royal Society B: Biological Sciences 283:20160333. https://doi.org/10.1098/rspb.2016.0333 DOI: https://doi.org/10.1098/rspb.2016.0333
Throop HL, Lerdau MT (2004) Effects of Nitrogen Deposition on Insect Herbivory: Implications for Community and Ecosystem Processes. Ecosystems 7. https://doi.org/10.1007/s10021-003-0225-x DOI: https://doi.org/10.1007/s10021-003-0225-x
Vanderplanck M, Leroy B, Wathelet B, Wattiez R, Michez D (2014) Standardized protocol to evaluate pollen polypeptides as bee food source. Apidologie 45:192-204. https://doi.org/10.1007/s13592-013-0239-0 DOI: https://doi.org/10.1007/s13592-013-0239-0
Vaudo AD, Patch HM, Mortensen DA, Tooker JF. Grozinger CM (2016) Macronutrient ratios in pollen shape bumble bee (Bombus impatiens) foraging strategies and floral preferences. Proceedings of the National Academy of Sciences 113:E4035. https://doi.org/10.1073/pnas.1606101113 DOI: https://doi.org/10.1073/pnas.1606101113
Vossler FG (2015) Broad Protein Spectrum in Stored Pollen of Three Stingless Bees from the Chaco Dry Forest in South America (Hymenoptera, Apidae, Meliponini) and Its Ecological Implications. Psyche: A Journal of Entomology 2015:1-7. https://doi.org/10.1155/2015/659538 DOI: https://doi.org/10.1155/2015/659538
Woodcock TS, Larson BMH, Kevan PG, Inouye DW, Lunau K (2014) Flies and Flowers II: Floral Attractants and Rewards. Journal of Pollination Ecology 12:63-94. https://doi.org/10.26786/1920-7603(2014)5 DOI: https://doi.org/10.26786/1920-7603(2014)5
Published
How to Cite
Issue
Section
License
Copyright (c) 2023 Luisa Carvalheiro, Dr. Maryse Vanderplanck, Dr. Mercedes Bustamante

This work is licensed under a Creative Commons Attribution 4.0 International License.











